AGES Radar for Infectious Diseases – 12 February 2026
Summary
Influenza activity is at a moderate level, and the positivity rate is falling significantly. The number of confirmed RSV cases has been rising since the end of December, and there has also been a significant increase in hospital admissions. SARS-CoV-2 is circulating at a low level.
Measles is no longer considered eliminated in Austria, and five other European countries have also had their elimination status revoked.
In several countries worldwide – including Austria – various infant formula products are currently being recalled after the toxin cereulide was detected.
India and Bangladesh have reported cases of the Nipah virus, prompting several countries in the region to step up health screenings at airports. In this month’s feature, we look at the hepatitis A virus and examine the outbreak situation in Austria and Europe in 2025.
In the news section, we report on the first detection of avian influenza in European dairy cows, the US’s final withdrawal from the WHO, and the end of the Marburg virus outbreak in Ethiopia.
Influenza activity is declining significantly. The number of influenza detections in the sentinel system is falling, and the peak of the flu season has passed. AGES wastewater monitoring also shows a significant decline, both in the measured values and in the number of sewage treatment plants with samples testing positive for influenza. Influenza detections have so far been almost exclusively attributable to influenza A. At the start of the season, circulation was predominantly characterised by the A(H3N2) subtype. In recent weeks, the proportion of the A(H3N2) subtype has decreased, whilst A(H1N1) has increased. The distribution is currently balanced. Influenza B has so far only been detected in isolated cases.
Hospital admissions for influenza have been falling since the start of the year: at the end of January, 305 admissions to general wards were recorded, compared with 882 four weeks earlier. As corrections and late reports are to be expected, the data may still change.
Influenza circulation is high in the European Union and the European Economic Area (EU/EEA). The peak appears to have passed in some countries, whilst in others influenza activity is approaching its peak. Hospital admissions have been declining overall since the start of the year. As in Austria, there is also co-circulation of influenza subtypes A(H3N2) and A(H1N1) at European level.
The best preventive measure against severe influenza is the annual flu vaccination. The currently circulating subclade K exhibits genetic changes compared to the A(H3N2) vaccine strain selected for vaccination. Initial data on vaccine effectiveness suggest that the seasonal vaccines available in the EU offer protection against infection with influenza A(H3N2). These results are consistent with findings from England. However, a definitive assessment of vaccine effectiveness can only be made retrospectively after the end of the season.
In Austria, vaccination against influenza (true flu) is recommended from the age of 6 months and is available free of charge to all age groups under the public vaccination programme.
Vaccination is particularly important for people at risk of severe disease and their close contacts/household contacts, as well as for people who are at increased risk of infection due to their circumstances (including pregnant women) or occupation. A nasal vaccine is available specifically for children. Further details can be found at www.impfen.gv.at/influenza and in the current Austrian Vaccination Schedule 2025/2026.
In episode 003 – Influenza & Co: How do I navigate the flu wave safely? of the AGES podcast “Mut zum Risiko”, infectious disease epidemiologist Fiona Költringer explains what the flu is all about and how best to protect yourself against it.
Since the end of December, the number of respiratory syncytial virus (RSV) cases detected in the sentinel surveillance system has been rising, particularly sharply over the last two weeks. The positivity rate has been above 10% since the end of January. Hospital admissions for RSV infections rose sharply at the end of January: with 139 admissions to general wards, there were more than twice as many in calendar week 5 as in the previous week. In wastewater, the trend is characterised by an upward trajectory.
In the rest of the EU and the EEA, RSV circulation is elevated and continues to rise. The season is therefore starting later than in the last two years.
Infants and young children, as well as people aged 60 and over, are at particularly high risk of developing severe RSV infection and requiring hospitalisation.
There is currently no authorised RSV vaccine for active immunisation in children. However, monoclonal antibodies are available for passive immunisation: Beyfortus (nirsevimab) is authorised and recommended for the prevention of lower respiratory tract RSV disease in newborns, infants and young children during their first RSV season, as well as in children up to 24 months of age who remain susceptible to severe RSV disease during their second RSV season. It will also be available in 2025/26 under the free childhood vaccination programme funded by the federal government, the Länder and social insurance. A vaccination for pregnant women is also authorised for the passive immunisation of children.
The RSV vaccine is recommended for adults aged 60 and over, as well as for people aged 18 and over who are at increased risk, to protect the vaccinated person against RSV infections/illnesses.
Further information on the vaccinations is available at impfen.gv.at/impfungen/rsv and in the current 2025/2026 vaccination schedule.
In the Sentinel system, the proportion of positive SARS-CoV-2 samples was low at around 5% in mid-January. The positivity rate has remained at this low level since the beginning of January. Wastewater monitoring has generally shown a downward trend over the last four weeks.
The number of hospital admissions due to severe acute respiratory infections caused by COVID-19 stood at around 330 per week in December and has been falling since the turn of the year. In calendar week 5, 95 admissions to general wards were recorded.
In the EU and the EEA, SARS-CoV-2 is circulating at low levels across all age groups. The impact on hospital admissions has so far been limited.
The COVID-19 vaccine is available free of charge in Austria and is recommended for everyone aged 12 and over who wishes to reduce the risk of a potentially severe course of the disease. Further information on the vaccine and its indications can be found in the Austrian Vaccination Schedule 2025/2026.
Austria is no longer officially considered ‘measles-free’. At the end of January 2026, the WHO announced that Austria had lost the measles elimination status it had achieved in 2015. This means that endemic transmission of measles is once again considered to have been ‘re-established’ in our country. This decision by the European Commission for Measles and Rubella Elimination (RVC – Regional Verification Commission) was based on the data submitted for the year 2024 as part of the annual status update.
In Austria, case numbers had risen sharply again since 2023. Following 186 cases in 2023, a total of 542 measles cases were recorded in Austria in the ‘record year’ of 2024. The following year, 2025, saw signs of a slight improvement, with 152 reported measles cases. Since the start of this year, two cases have been confirmed so far (as of 11 February 2026).
The RVC’s decision also took into account indicators describing the quality of the surveillance system, laboratory diagnostic procedures and the implementation of the vaccination programme in Austria. One of the core strategies for the elimination of measles and rubella is the nationwide “achievement and maintenance of high vaccination coverage rates >95% with two doses of a measles and rubella vaccine”. Austria is far from achieving this.
The Vienna University of Technology evaluates vaccination coverage rates for Austria annually on behalf of the Ministry of Health. This has shown that the target of 95% vaccination coverage with two doses has still not been achieved for most age groups. Vaccinations are also taking place later than recommended. The low vaccination coverage rates particularly affect the 2019 and 2020 cohorts, where gaps in vaccination coverage have likely arisen due to lockdowns and other measures to reduce contact during the COVID-19 pandemic. Given that children in this age group are starting nursery school, it is particularly important to check their vaccination status and catch up on any missing doses.
Along with Austria, five other countries in the WHO European Region have lost their ‘measles-free’ status: the United Kingdom, Spain, Armenia, Azerbaijan and Uzbekistan. Measles is considered endemic in 13 countries in the WHO European Region, including the EU Member States Germany, Italy, France, Poland and Romania.
In 2025, the USA recorded 2,267 confirmed cases of measles, the highest number in over 30 years. Due to the outbreaks reported in the USA since January 2025, the relevant commission of the Pan American Health Organization (PAHO) plans to review the USA’s measles elimination status, together with that of Mexico, in April 2026.
Preliminary data for 2025 show a significant decline in reported measles cases in EU and EEA countries compared to 2024. However, these figures are still twice as high as those reported for 2023. The number of measles infections usually peaks in late winter and early spring, so the ECDC now recommends checking your own measles vaccination status.
The measles-mumps-rubella (MMR) vaccine is available free of charge in Austria for all age groups at all public vaccination centres and from all registered doctors. Missing vaccinations can and should be caught up on at any age. Further details can be found at impfen.gv.at/impfungen/masern-mumps-roteln or in the Austrian Immunisation Schedule 2025/2026.
European Regional Verification Commission for Measles and Rubella Elimination (RVC)
In several countries around the world – including Austria – various baby food products are currently being recalled. The reason for this is the detection of cereulide, a toxin produced by the bacterium Bacillus cereus.
The cereulide toxin causes vomiting, abdominal cramps and nausea, and, less commonly, watery diarrhoea. The symptoms usually subside within 24 hours. In very few cases, severe forms of the condition may occur. Symptoms of cereulide poisoning usually appear very quickly after consuming contaminated infant formula: within 15 minutes to 6 hours. The effects of potential exposure to or ingestion of the toxin range from mild to moderate, depending on the child’s age. Newborns and infants under six months are more likely to develop symptoms and may even suffer complications such as dehydration, electrolyte imbalances, etc. Therefore, the overall risk for infants under one year of age in the EU and the EEA is assessed as moderate in this case. (ECDC, as of 06/02/2026)
An overview of the batches affected in Austria to date can be found under Infant formula: Overview of all recalls – AGES
The ECDC has received several reports of diarrhoea in infants following consumption of the products. The analysis of cereulide toxins in stool samples is not a method available for routine diagnostic purposes in clinical microbiology laboratories. To date, Belgium is the only EU country in which five infants have tested positive for the toxin. All five had consumed infant formula from a recalled batch and have made a full recovery. The infant formula consumed tested positive in three cases; two samples are currently being examined at the reference laboratory. Other countries, such as France, Denmark, Spain and the United Kingdom, have also reported some cases of diarrhoea in infants; however, a link to the consumption of recalled products has not yet been established. (ECDC, as of 06/02/2026)
On 2 February 2026, the European Food Safety Authority (EFSA) published a rapid risk assessment on cereulide. Taking into account a safety factor of 100 and an additional safety factor of 3 for infants, an Acute Reference Dose (ARfD) of 0.014 µg of cereulide per kilogram of body weight per day has now been established for the first time. Cereulide concentrations exceeding 0.054 μg/L in infant formula and 0.1 μg/L in follow-on formula could therefore exceed safe levels. At the end of January, a cereulide concentration was detected in infant formula in Austria for the first time which, according to the new EFSA assessment, must be classified as harmful to health, as the ARfD is exceeded several times over. This product is among those that have already been recalled by the manufacturer.
On 3 February 2026, Bangladesh reported a case of Nipah virus (NiV) infection from a region in the west of the country. The patient had developed a fever and neurological symptoms on 21 January; a week later, she was referred to a tertiary hospital, where she died on the same day. On 29 January, the NiV infection was confirmed by laboratory tests. The patient had no travel history, but had consumed raw date palm sap on several occasions prior to falling ill, a source of infection previously described on multiple occasions during past outbreaks in the country. A total of 35 contacts identified during the investigations are currently being monitored; no further cases have been detected in Bangladesh to date. (WHO, as of 6 February 2026)
Shortly before this, on 26 January 2026, the Indian authorities reported two laboratory-confirmed cases of NiV infection in the state of West Bengal, in the north-east of the country and bordering Bangladesh to the west. The two affected individuals are nurses at a hospital in Barasat. They had developed typical symptoms of a severe NiV infection at the end of December and were admitted to hospital in early January. Both are receiving inpatient treatment (WHO, as of 30 January 2026). A total of 196 close contacts were identified and placed in quarantine. All close contacts tested negative for NiV (ECDC, as of 6 February 2026).
Several countries in the region, including Thailand, Nepal and Cambodia, have stepped up health and symptom screenings at international airports in light of the current regional situation.
At a global level, the WHO currently classifies the public health risk as low. According to the ECDC’s assessment, the current risk to the European population is considered very low.
The Nipah virus is a zoonotic virus belonging to the Henipavirus family. Fruit bats of the genus Pteropus are considered to be the natural reservoir. The virus is transmitted through contact with infected animals or through food contaminated by the saliva, urine or other excretions of infected animals. Human-to-human transmission is also possible through close contact with an infected person. Previous outbreaks have been linked, among other things, to the consumption of contaminated date palm sap. The virus has a high case fatality rate, which is reported to be between 40% and 75%, depending on local conditions regarding early detection and medical care. Currently, no specific antiviral treatment or vaccine is available. However, work is underway: at the end of December, the University of Oxford launched the world’s first Phase II trial of a NiV vaccine.
The first known Nipah outbreak was described in 1998 in Malaysia and Singapore in connection with infected pigs. At that time, 269 people fell ill, of whom 101 died.
NiV is included on the WHO’s list of prioritised pathogens (R&D Blueprint), which pose a major public health risk due to their epidemic potential or the current lack of adequate countermeasures. In the AGES-Radar issue of 11 December 2025, we explored ‘Disease X’ and these prioritised pathogens in more detail in our ‘Topic of the Month’.
Nipah virus infection – Bangladesh
Nipah virus disease cases reported in West Bengal, India: very low risk for Europeans
| Feature | Description |
|---|---|
| Occurrence | Found worldwide, particularly in regions with poor hygiene conditions. Rare in Austria, mainly associated with travel |
| Pathogen | Hepatitis A virus |
| Reservoir | Mainly humans |
| Route of transmission | Either direct faecal-oral (especially through close contact) or indirect faecal-oral (via contaminated food or drinking water, but also surfaces and objects). Asymptomatic cases, often children, can also be a source. |
| Incubation period | Average 1 month, range: 15–50 |
| Duration of infectiousness | At the earliest 2 weeks before and up to 1 week after the onset of symptoms; young children can shed the virus for up to 6 months. |
| Clinical symptoms | Initially non-specific symptoms (fever, fatigue, loss of appetite and nausea), followed frequently by upper abdominal pain on the right side, vomiting and elevated liver enzymes; often accompanied by itching and jaundice. Additionally, enlargement of the liver and, occasionally, rashes. In children under six years of age, the disease often has a subclinical or asymptomatic course. Fatal outcomes are rare. |
| Risk groups | Children and travellers to high-endemic areas, contacts, people who come into occupational contact with faecal matter, and people who use intravenous drugs. |
| Vaccination | Recommended for certain risk groups or as post-exposure prophylaxis following contact |
Hepatitis A: an introduction
Hepatitis A has been known since ancient times and is widespread worldwide, although the incidence varies greatly between regions. High incidence rates are found in areas with poor hygiene conditions, particularly poor water supply, and therefore mainly in low- or middle-income countries. In such high-endemic areas, infection usually occurs during childhood; in Africa and Asia, there are regions where over 90% of the population has serologically proven contact with the hepatitis A virus. In industrialised countries, i.e. low-endemic areas, this figure stands at approximately 33%. China, some countries in South America, Central and South-East Asia, and the Middle East are classified as intermediate-endemic areas. In these intermediate-endemic areas, as well as in low-endemic areas, hepatitis A outbreaks can occur under certain circumstances.
Hepatitis A is caused by the hepatitis A virus (HAV). Despite the similar names, this virus is not related to other hepatitis viruses. It belongs to the hepatoviruses, which, like polioviruses, rhinoviruses or Coxsackie viruses, belong to the Picornaviridae family. The naming of hepatitis viruses is based on the clinical presentation of hepatitis, or inflammation of the liver, and has nothing to do with genetic relatedness. HAV was first visualised under an electron microscope in 1973. In humans, it can be divided into three genotypes (I–III) with subtypes A and B; genotypes IV–VI affect monkeys. All genotypes belong to a single serotype, which is relevant for the immune response: regardless of the genotype, the same neutralising antibodies are produced and lifelong immunity is conferred. Like other non-enveloped viruses, HAV is highly resistant to environmental conditions and can remain infectious for weeks in food, soil, or in fresh and salt water. It is also resistant to temperature fluctuations; for example, it can survive freezing and temperatures of up to 60 degrees Celsius. It also has reduced sensitivity to soap. Only specific disinfectants labelled ‘virucidal’ or specifically tested against HAV effectively kill the virus.
HAV is excreted in the stool of infected individuals, whether symptomatic or asymptomatic; the route of transmission is faecal-oral. Transmission from person to person can occur either directly through close contact or sexual contact, or indirectly via contaminated water, e.g. raw seafood or frozen berries, food, surfaces or objects. Consequently, the spread of the disease is closely linked to hygiene conditions. Transmission via blood/blood products or through the sharing of blood-contaminated needles has also been reported. Hepatitis A is highly contagious; it is thought that even a small dose of 10 to 100 viral particles is sufficient to cause infection. Initial symptoms appear approximately one month after exposure, although the incubation period can range from two weeks to 50 days. Those affected who develop symptoms are contagious from two weeks before the onset of symptoms until approximately one week after symptoms begin. Contagiousness is highest in the period around the onset of symptoms. Children play a particular role here, as they often show no symptoms and can shed the virus over a long period (up to six months). All these characteristics – high environmental resistance, low infectious dose, contagiousness before symptom onset, asymptomatic carriers – contribute to rapidly spreading outbreaks.
The disease itself begins with non-specific, flu-like symptoms such as fever, fatigue, loss of appetite and nausea; subsequently, inflammation of the liver becomes apparent, with discomfort in the upper right abdomen, elevated liver enzymes or symptoms such as itching and jaundice. In addition, the liver may become enlarged and transient rashes may occur. The disease usually resolves on its own. Although recovery can take months, it does not lead to chronic liver inflammation. Mortality is low, at approximately 0.1 to 0.3 per cent; the risk increases with age and pre-existing liver conditions.
Children and travellers to high-endemic areas, people who have been in contact with hepatitis A cases, those who come into occupational contact with faecal matter (e.g. in laboratories, sewer workers), and people who inject drugs are at increased risk of hepatitis A infection.
In Austria, vaccination is not currently recommended for children in general, but only for specific risk groups who are at increased risk of hepatitis A (travellers; healthcare workers; men who have sex with men (MSM); social care workers) or for a severe course of the disease (chronic liver disease, chronic intestinal diseases), or who are at increased risk of spreading the disease (staff in food businesses or communal catering). Vaccination is possible from the age of 1 using an inactivated vaccine and is administered either as two doses or as a combined vaccine against hepatitis A and B in three doses. Immunity lasts for a long time after vaccination; a booster is generally not necessary. For further details on vaccination recommendations, see the chapter on Hepatitis A under ‘Travel/Indicated Vaccinations’ in the Austrian Immunisation Schedule 2025/2026.
Hepatitis A in Austria
| Hepatitis A, Austria 2025 |
|---|
| Total number of cases 249 |
| Incidence 2.71 / 100,000 |
| Median age (IQR) 35 (23–49) |
| Gender (M:F) 154:84 |
| Hospitalised 127 (51%) |
| Deaths 4 (1.6%) |
Austria is classified as a low-endemic area, primarily due to widespread and adequate sanitation; infections are frequently acquired abroad. As a rule, around one third of cases are travel-associated. In recent years, the incidence of hepatitis A has been less than one case per 100,000 inhabitants; in absolute terms, this amounts to between 30 and 80 cases per year (Fig. 1).
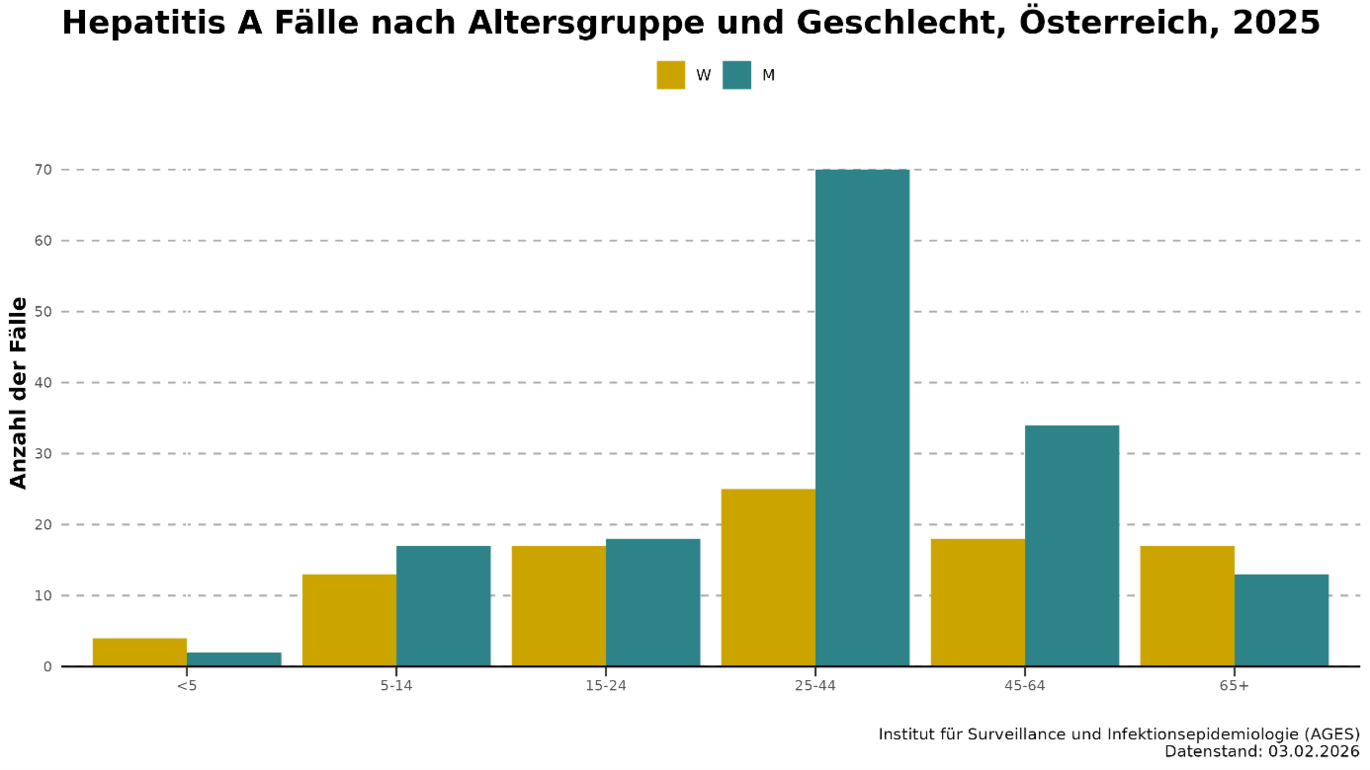
In Austria, men and middle-aged people are slightly more frequently affected by hepatitis A, whereas children rarely contract the disease (Fig. 2). Hospitalisation is common among reported cases; around half of those affected require hospital admission. Admissions to intensive care units (0 to 2 per year) and deaths (0 to 1 per year) are rare.
The years 2016/2017 and 2025 are exceptions to the normally low incidence of hepatitis A (see Fig. 1). From summer 2016 through to 2017, there was a global (primarily in Europe and America) hepatitis A outbreak: the incidence in Europe doubled compared with previous years, with Slovakia, Romania and Bulgaria being the hardest hit. A total of 26,294 cases were reported in the EU in 2017, with men aged between 25 and 44 being the most affected. The causative viruses could be attributed to three different strains. The outbreak was mainly driven by person-to-person transmission; food played a minor role, and MSM were disproportionately affected. In Austria, 94 and 242 cases of hepatitis A were reported in 2016 and 2017 respectively, with incidence rates of 1.07 and 2.74 per 100,000 inhabitants. Travel-associated cases accounted for less than a quarter of the total in those years.
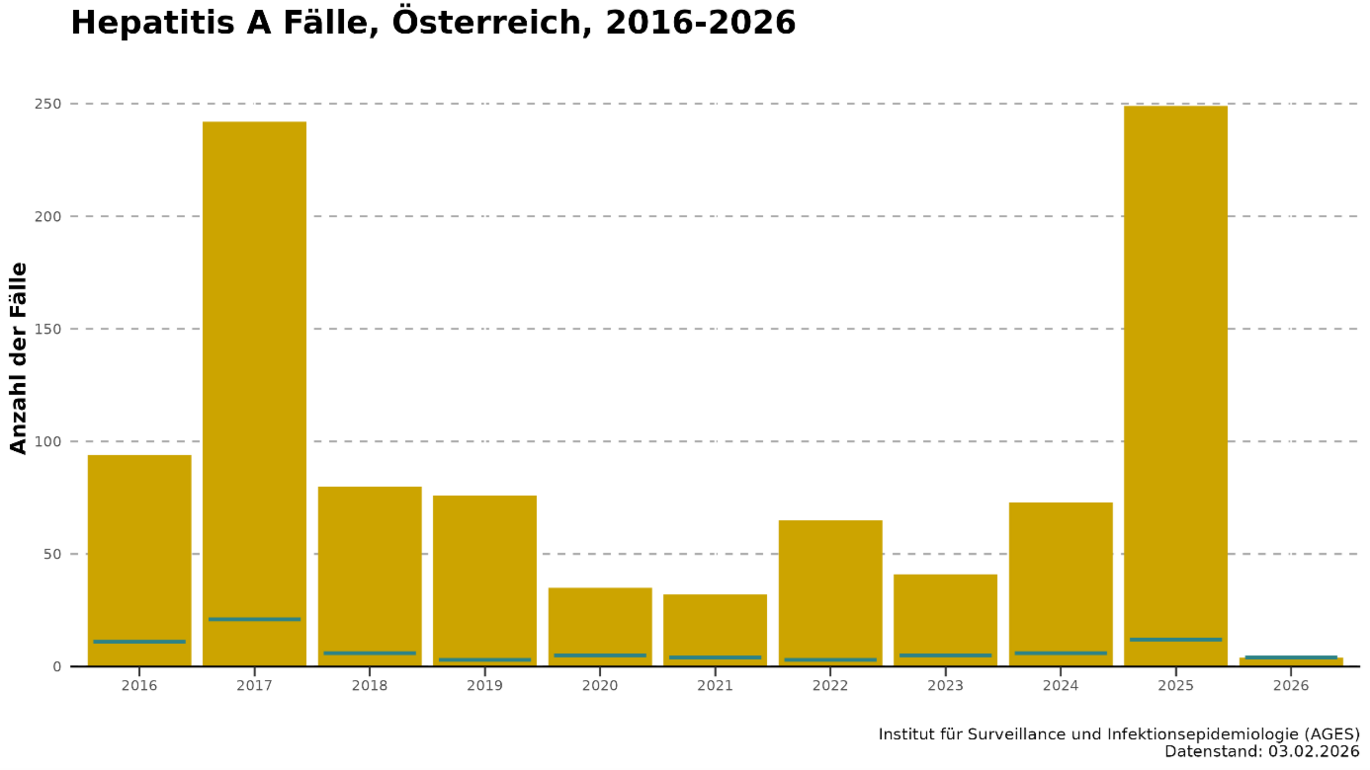
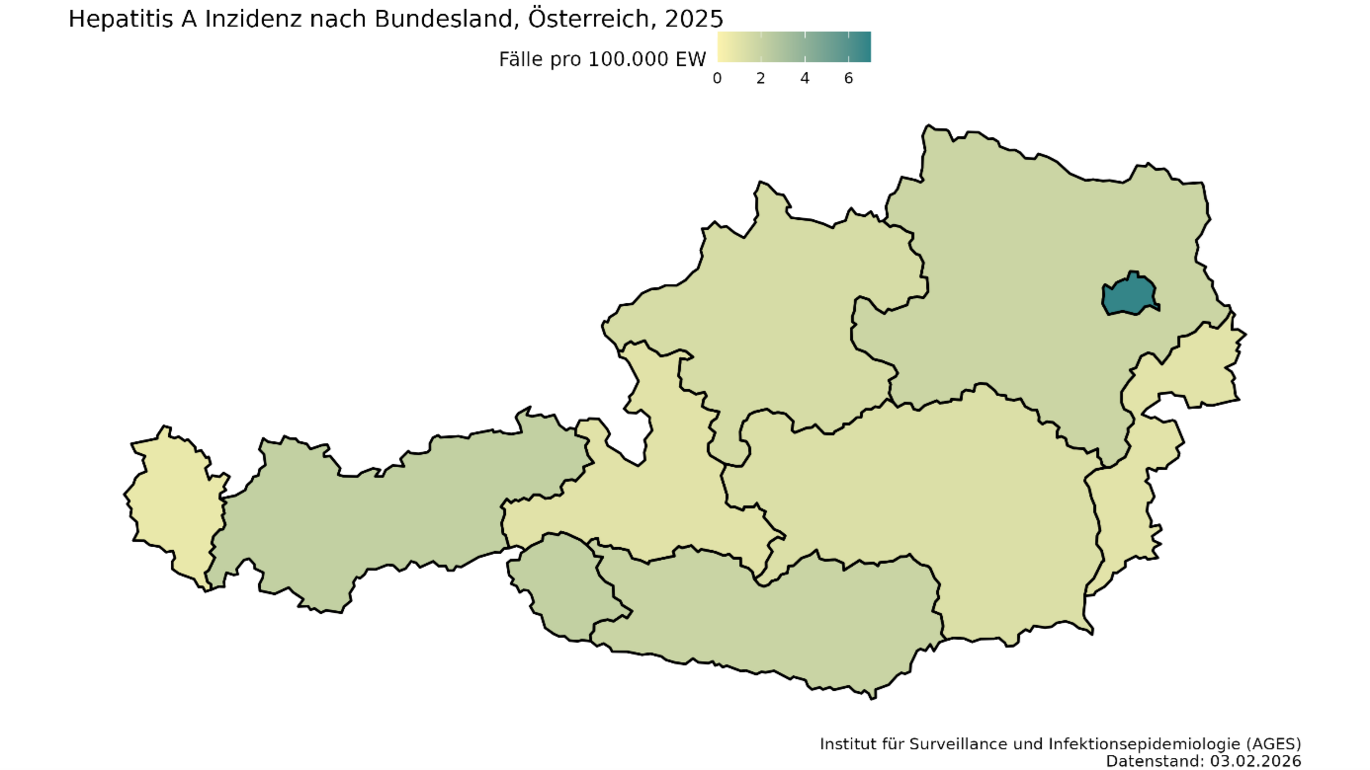
In 2025, Austria saw another sharp rise in hepatitis A cases, particularly in Vienna (see Fig. 3). By March, 32 cases had already been reported; by May, the number of infections had already exceeded the total for the whole of previous years. A total of 249 cases were reported, with an incidence of 2.71 cases per 100,000 inhabitants. Molecular biological analysis of the hepatitis A viruses by the National Reference Centre also revealed that, in many cases, the infection was caused by one of the two circulating HAV strains. Both HAV strains belong to genotype IB and are closely related. To date, a total of 131 cases have been linked to the two outbreaks, three of which are from 2023 and 2024 respectively. Five patients (3.8%) died as a result of the outbreak. 42% had to be admitted to hospital for treatment, and four patients were treated in the intensive care unit. The median age of those affected was 37 years, and 76% were men. The outbreak investigation ruled out contaminated food as a cause. As was the case in 2017, the outbreak appears to have spread mainly through direct person-to-person transmission. A strikingly high proportion of the cases surveyed involved people experiencing homelessness (31%) and people who inject drugs (17%). In just under a third of cases, infection is suspected to have occurred abroad, with various countries being cited. In one-fifth of the cases surveyed, there had been either contact with people suffering from hepatitis A or diarrhoea and/or high-risk sexual contact prior to infection.
It became known via the European Surveillance Portal and subsequently through a Rapid Risk Assessment by the ECDC that Austria is not the only country affected by a rise in hepatitis A cases. The ECDC reported in November 2025 that a total of over 6,000 hepatitis A cases had been reported across European countries. Slovakia, with 2,482 cases, the Czech Republic with 2,310 cases and Hungary with 1,548 cases account for the largest share. The Austrian cases are linked to those in the three neighbouring countries; they share the same outbreak strains. In all countries, groups of people living in poor hygienic conditions or with insufficient access to the healthcare system are disproportionately affected. In addition to homeless people and those who inject drugs, a particularly high proportion of Roma communities are affected in the neighbouring countries. In Hungary and Slovakia, unlike in Austria and the Czech Republic, more children than adults are affected. In Slovakia, the outbreak is thought to have begun as early as late 2022 and reached its peak in 2023/2024, whereas in the other affected countries it did not peak until late 2024/early 2025. Isolated cases also occurred in Germany, Sweden and the United Kingdom.
In all countries, including Austria, vaccination and information campaigns were carried out among the affected populations as well as among care and treatment staff. In Austria, case numbers have been declining since a peak in the summer of 2025, with the last case reported in early December. It is still unclear how the outbreak will develop in an international context.
On 23 January 2026, the Dutch authorities reported the detection of antibodies against the avian influenza virus A(H5N1) in the milk of a dairy cow. The cow had previously shown signs of mastitis (inflammation of the udder) and respiratory symptoms. Milk from the affected cow did not enter the food supply chain, and foodborne transmission is not currently considered a risk.
Since then, antibodies against the influenza A(H5N1) virus have been found in four further cows at the same dairy farm. Antibodies indicate a past infection, making these the first cases of avian influenza in dairy cows in Europe. The authorities became aware of the farm after the avian influenza virus was detected in a cat in the vicinity of the farm. None of the people exposed on the farm have developed symptoms, and no antibodies have been detected either. Further investigations are ongoing.
To date, there have been no confirmed cases of Influenza A(H5N1) in humans in the EU or the EEA. In the USA, however, there have been instances of transmission to humans in connection with infections in dairy cows. These were, however, rare and mild in nature. The ECDC’s risk assessment therefore remains unchanged: the current risk is assessed as low for the general population, and as low to moderate for people with occupational exposure (e.g. poultry farm workers) or other exposure to infected animals or contaminated environments.
A vaccine against avian influenza is available free of charge at certain facilities in Austria. Due to the epidemiology, specific infection patterns and low probability of infection, vaccination against avian influenza is currently not recommended or planned for the general public. Vaccination is recommended for individuals who may have (close) contact with infected animal populations, particularly bird populations. Further information on vaccination centres can be obtained from the relevant provincial health authorities. The vaccination recommendation for avian influenza (bird flu) is available online at sozialministerium.gv.at/impfplan.
You can also find all information on bird flu in episode 10: ‘The chickens aren’t laughing – bird flu in the spotlight’ of our AGES podcast ‘Mut zum Risiko’ with expert Irene Zimpernik.
On 26 January 2026, the Ethiopian Ministry of Health officially declared the country’s first-ever Marburg virus outbreak to be over, after no new cases had been reported for a period of 42 days – in accordance with the WHO criteria for outbreak control.
On 14 November 2025, the first cases of Marburg virus disease were confirmed in Ethiopia. A total of 19 cases were recorded during the outbreak, 14 of which were laboratory-confirmed. Nine of the confirmed cases and the five probable cases were fatal (WHO, as of 26 January 2026).
On 22 January 2026, the US formally withdrew from the WHO. In a statement dated 24 January 2026, the WHO acknowledged the withdrawal and addressed the US press release regarding the withdrawal.
The US had been a founding member since the WHO was established in 1948 and was the largest contributor.
For a deeper insight into the WHO’s powers and resources: in the AGES-Radar issue of 20 February 2025, we wrote about the WHO and the US’s planned withdrawal in our ‘Topic of the Month’ section.
The 2026 Winter Olympics will take place in Italy from 4 February to 22 February, bringing together visitors from all over the world. The ECDC has therefore published health advice. Travellers should check their vaccination status, particularly against measles, as outbreaks have been reported worldwide and in Italy. It is also recommended to protect oneself against respiratory infections such as flu, COVID-19 and RSV, and to observe basic hygiene measures. For food and water safety, advice includes thorough cooking, hand washing and caution regarding food left unrefrigerated for long periods. People are also urged to protect themselves against sexually transmitted infections.
The Lombardy region has set up a dedicated website for the 2026 Winter Olympics providing information on emergency services, medical support and medicines.
The ECDC is monitoring this mass event as part of its epidemiological surveillance activities, in close cooperation with the Italian National Institute of Health and other partners.
Case numbers for notifiable diseases under the Epidemics Act; the figures shown are for the previous month and, for the period from the start of the year to the end of the previous month, the figures for the current year, the previous year, and the median of the last five years for comparison (Epidemiological Reporting System, as at 11 March 2026).
a Diseases are assessed in accordance with the case definition. Diseases for which a case definition exists are shown, with the exception of transmissible spongiform encephalopathies. As a rule, confirmed and probable cases are counted. Changes may still occur due to late reporting or subsequent entries.
b Bacterial and viral food poisoning, in accordance with the Epidemics Act.
c Invasive bacterial disease, in accordance with the Epidemics Act.
d Includes only cases involving pneumonia.
e Due to the lack of a case definition prior to 2025, only cases from 2025 onwards are shown; the median is also calculated only from 2025 onwards.
f Mpox has been a notifiable disease since 2022; the median is calculated only for the years in which it is a notifiable disease.
Last updated: 22.05.2026
automatically translated