If a child shows typical symptoms – especially vomiting – within six hours of consuming infant formula, please contact your local food safety authority, stating the product name. The authority will then initiate further investigations. If symptoms persist or are severe, please contact your paediatrician immediately.
In addition to Austria, Belgium, Denmark, France, Luxembourg, Spain and the United Kingdom have also reported cases that are currently being investigated. The joint assessment by the European Centre for Disease Prevention and Control (ECDC) and the European Food Safety Authority (EFSA) shows that most of the reported cases had mild gastrointestinal symptoms. However, some hospitalisations due to dehydration have also been reported.
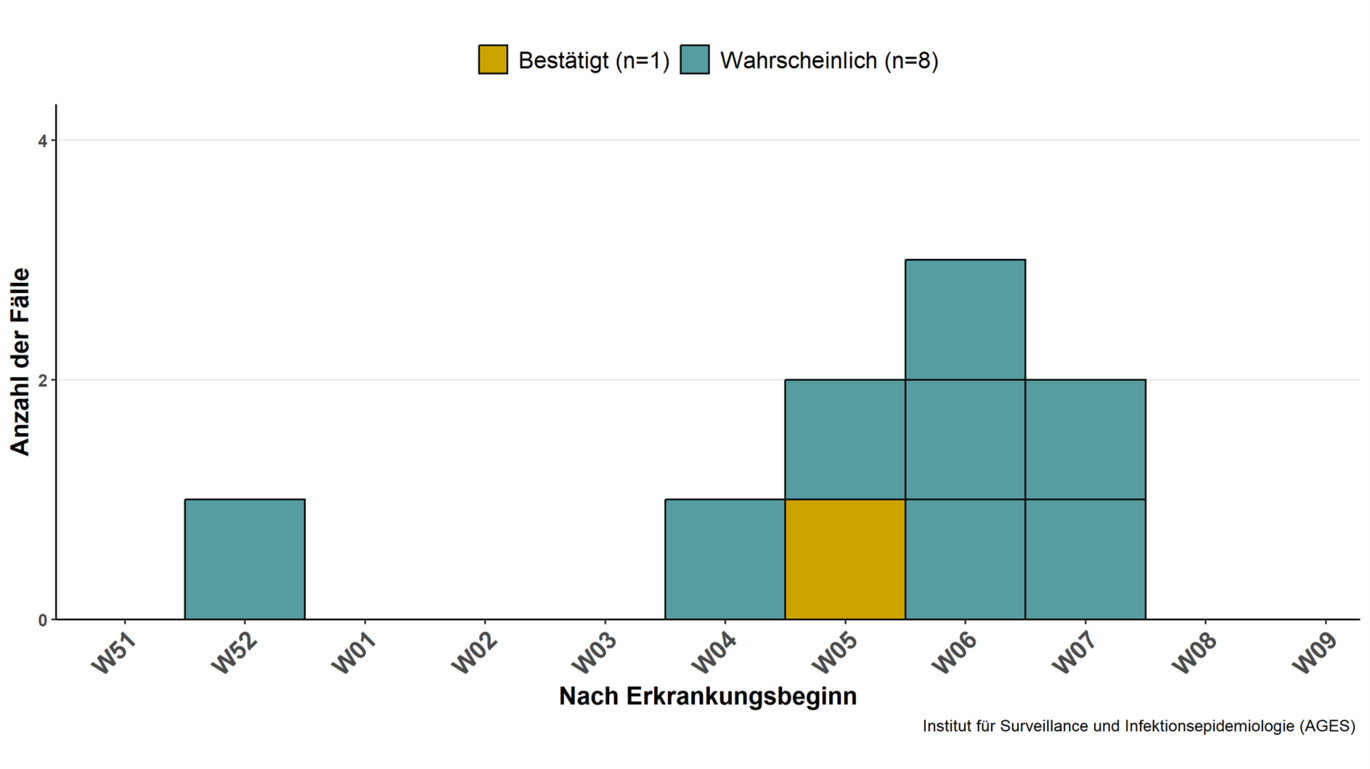
Case definitions
Confirmed case
A confirmed case of intoxication by the emetic Bacillus cereus toxin (cereulide) is defined as a child under 36 months of age with gastrointestinal symptoms, particularly vomiting, in which the onset of illness occurs within 6 hours of consuming infant formula and in which cereulide has been detected in a stool sample or in the batch of infant formula consumed at a concentration exceeding the EFSA reference value and the corresponding food sample has been classified as "unsafe for human consumption" according to expert opinion.
Probable case
A probable case of intoxication by the emetic Bacillus cereus toxin (cereulide) is defined as a child under 36 months of age with gastrointestinal symptoms, in particular vomiting, in which the onset of illness occurs within 6 hours of consuming infant formula from a batch known to contain cereulide, or in which cereulide has been detected in a batch of the infant formula consumed at a concentration below the EFSA reference value.
Investigations of infant formula
As of 25 February 2026, AGES has tested 145 samples of infant formula from 9 different manufacturers for cereulide as part of the priority actions and other official controls commissioned by the Ministry of Health:
- 120 samples contained no detectable amounts of cereulide
- Cereulide was detected in 25 samples. Most of the values are well below the level determined by the EFSA at which health effects cannot be ruled out. In all cases, these are products from manufacturers who have already recalled their products.
Further samples are currently being tested.
Cereulide analysis
Contaminated ARA oil is considered to be the cause of the contamination of infant formula with the bacterial toxin cereulide. This oil is added to infant formula as a source of polyunsaturated fatty acids, as these fatty acids are important for the development of the nervous system and vision.
This ARA oil is present in the powdered infant formula in the form of tiny capsules. This means that the cereulide is also enclosed in these capsules. It has been shown that, with the standard testing method used to date, there are significant differences in the analysis results depending on whether the powder or the ready-made infant formula is tested: in the finished bottle, the cereulide concentrations were significantly higher than those measured in the powder.
This is not the case with the AGES testing method. Many years ago, AGES established an analytical method for determining cereulide, which is used on an ongoing basis to test various foods. With this method, it makes no difference whether the powder or the ready-made food is tested: the cereulide concentrations determined are the same in both cases.
The basis for this lies in the preparation of the samples, which makes it possible to extract the substance to be tested from the food in its entirety. Another advantage of the AGES method is that it allows even less sensitive equipment to detect small amounts of cereulide.
Thanks to this method and close cooperation with food authorities, Austria was the first country in the EU to discover samples contaminated with cereulide in the course of official food inspections. At the end of January, the European Commission therefore made the AGES method for cereulide analysis available to all Member States in order to establish an alternative to the standard testing method if necessary.
All recalls at a glance